- Статьи
- Science and technology
- The enzyme of truth: scientists have uncovered the secret weapon of the indestructible tuberculosis bacillus
The enzyme of truth: scientists have uncovered the secret weapon of the indestructible tuberculosis bacillus

Russian scientists were the first to uncover the secret weapon of tuberculosis bacillus: they discovered an enzyme that works on the principle of a "molecular carousel." It helps mycobacteria to enter a state of "hibernation" and maintain resistance to antibiotics and the immune system. The discovery could be the key to creating fundamentally new drugs against tuberculosis, which, according to the latest published WHO data, affects over 10 million people worldwide. This is a promising target for pharmacology, but it may take 10-15 years before a potential drug is put on the market, experts interviewed by Izvestia warned.
The mechanism of the molecular carousel
Scientists have discovered an enzyme that helps mycobacteria— the causative agents of tuberculosis, survive the attack of the immune system and the effects of antibiotics. It is responsible for creating molecules that increase the rigidity of the bacterial membrane and slow down the metabolism in the cell. The discovery made by scientists from the Federal Research Center "Fundamental Foundations of Biotechnology" of the Russian Academy of Sciences and the Institute of Bioorganic Chemistry named after academicians M.M. Shemyakin and Yu.A. Ovchinnikov of the Russian Academy of Sciences will be used in the development of new drugs.
According to WHO statistics, 10.7 million people worldwide were diagnosed with tuberculosis in 2024. The causative agents of the disease are mycobacteria (Mycobacterium tuberculosis), which, after entering the body, are able to enter a state of prolonged "hibernation". In it, the cells do not multiply, but become extremely resistant to adverse factors, including the action of antibiotics and the immune response. As a result, even after seemingly successful treatment, the disease can recur years later due to the reactivation of dormant mycobacteria.
Studies have shown that a special substance, coproporphyrin tetramethyl ether, accumulates in mycobacterium cells at rest. Coproporphyrin itself is a molecule with a ring structure, and the addition of four methyl groups to it leads to the formation of ether, which accumulates in the bacterial cell wall. It is this compound that increases the resistance of mycobacteria to adverse factors, including the effects of the immune system and oxidative stress. At its high concentration, the activity of bacterial respiration decreases, which, along with other metabolic changes, contributes to the transition of cells to a state of "hibernation". However, until recently, it remained unclear how such a chemical transformation occurs.
Russian scientists have identified an enzyme responsible for the conversion of coproporphyrin into its essential form. To do this, they analyzed protein databases and selected the most likely candidate enzymes present in mycobacteria. Next, the researchers obtained experimental strains: some with increased production of this enzyme, others without the ability to synthesize it, which confirmed its key role in this process.
Bacteria with an excess of the enzyme accumulated nine times more ether than normal and were much better able to survive conditions that mimic the attack of the immune system in the human body and the effects of antibiotics. Mycobacteria without this enzyme synthesized protective molecules worse, which reduced their survival rate.
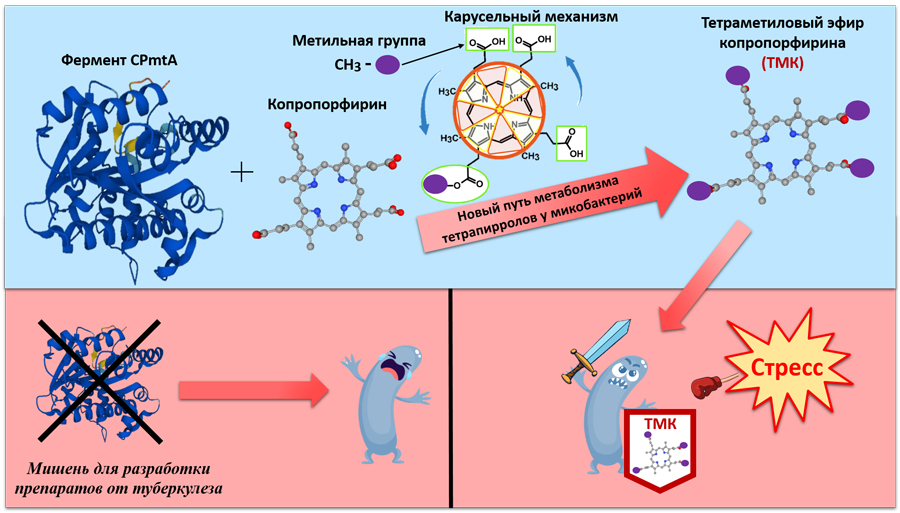
Using computer modeling, the authors found out that the open enzyme operates by an unusual mechanism similar to a "molecular carousel." Coproporphyrin enters the active center of the enzyme and, being in it, changes its orientation (rotates), as a result of which four methyl groups are "hung" on it. This makes it possible to synthesize tetramethyl ether directly without intermediates and thereby increase the conversion efficiency.
The resulting ether is embedded in the membrane of the microorganism, increases its rigidity and slows down the metabolic processes. All this serves as a signal for switching the cell to an energy-saving mode, which contributes to long-term survival in adverse conditions. It is in this state that mycobacteria "hide" from the immune system and drugs in the human body.
— Since the described enzyme is active precisely during the transition of mycobacteria to a dormant state, it can serve as a target for the development of drugs for tuberculosis. So, if you block it, the bacterium will not be able to go into a dormant state and will become sensitive to the action of the immune system and antibiotics," said Margarita Shleeva, project manager, Doctor of Biological Sciences, Head of the Laboratory of Stress Biochemistry of Microorganisms at the Institute of Biotechnology of the Russian Academy of Sciences.
In the future, scientists plan to identify all the enzymes involved in this new biochemical pathway in mycobacteria.
Prospects in clinical medicine
Tuberculosis bacillus is able to build a molecular "armor" around itself, which makes it almost invulnerable to immunity and antibiotics, said molecular biologist Arina Kholkina.
— Scientists have found an "engineer" enzyme that creates the material for this armor — tetramethyl ether of coproporphyrin. The bacterium quickly "puts on its armor" and goes into hibernation for years. If you block this enzyme with a drug, the wand will remain unarmored and become vulnerable to conventional drugs and immunity," the specialist told Izvestia. — This is a very promising target for pharmacology: the carousel mechanism is unique and absent in humans, which means that the inhibitor will be safe. Such a remedy will deprive the bacterium of its main trick (the ability to hide), shorten the duration of therapy and help prevent relapses and latent infections.
Multidrug—resistant tuberculosis is an increasing global problem, so any mechanism of action on dormant forms is of strategic interest, added Marina Chumakova, a leading market expert at NTI Helsnet.
— Most of the existing antibiotics (isoniazid, rifampicin) attack actively dividing cells. Dormant mycobacteria do not respond to them, which explains the recurrence years later. An inhibitor of this enzyme theoretically attacks a condition that is now virtually inaccessible for therapy," she noted.
At the same time, the expert emphasized, the path from scientific discovery to the creation of a drug remains a long one: the realistic horizon of its appearance is estimated at 10-15 years.
The results of the study, supported by a grant from the Russian Science Foundation (RSF), are published in the International Journal of Biological Macromolecules.
Переведено сервисом «Яндекс Переводчик»






